A prospective observational study to evaluate intraoperative hyperspectral imaging for real-time quantitative fluorescence-guided surgery of low-grade glioma.
Official accronym: Neuro-qFHSI
IRAS: 307291. REC reference: 22/LO/0299.
ClinicalTrials.gov Identifier: NCT05397574
Award ID: WT223880/Z/21/Z
Contact the NeuroPPEye team using our contact form.
The study
A study to demonstrate that a novel, light based, imaging system could be used in neurosurgical procedures in the future with a view to improving outcomes
The key points
- Will not affect your operation or outcome
- Study to assess whether the system is capable of obtaining helpful images for surgical guidance in the future
- May add a maximum of 15 minutes to your operation time in order to obtain these images but no other aspect of the procedure will be carried out any differently
- No radiation, the system uses safe light only
- No additional follow-up visits or contact required

More details can be read in our Plain English Summary and Scientific Abstract.
Patient and public involvment
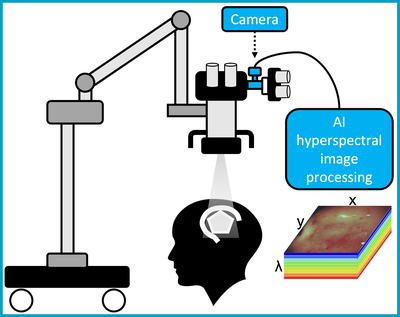
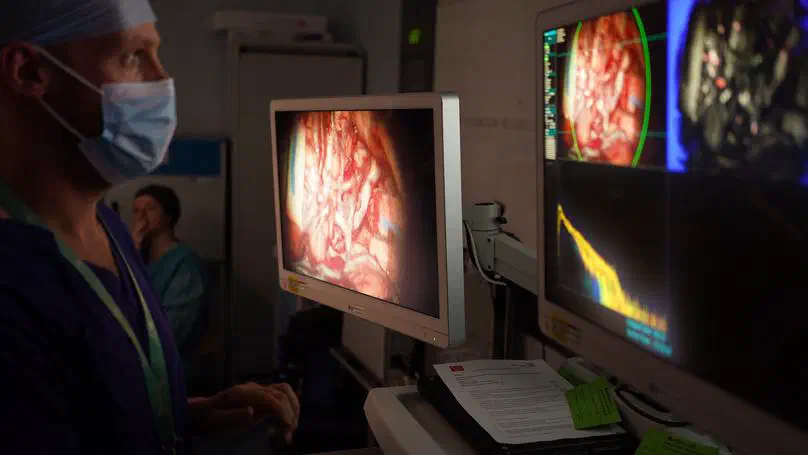
The purpose of this study is to obtain images of brain tumours during surgery using a new type of surgical camera. This is called a “hyperspectral” camera. Normal cameras work by recording visible light - and using that to generate an image. Hyperspectral cameras record this information, but also record wavelengths of light (or spectra) that we cannot see. Recent studies have suggested that when this extra information is processed by additional software, it may provide extra information to the surgical team, for example information regarding blood flow or the location of tumour boundaries.
In certain types of brain tumour surgery, a type of medication called “Gliolan” or “5 ALA” or “pink drink” is given before surgery. This includes a compound that builds up in tumour cells and makes them glow (or fluoresce) pink when blue light is shined on them. This helps the surgeon remove parts of the tumour that can otherwise be difficult to identify and is reliant on enough pink light being emitted for the surgeon to see. Recent studies have suggested that very low levels of fluorescent light, which may not be visible to the naked eye, could be detectable using hyperspectral imaging. Some studies also suggest it can quantify exactly how much light is being emitted in real time – potentially giving surgeons live information about the tumour type and boundaries.
The study will assess how the information obtained from hyperspectral images during surgery matches the removed tissue. Data will also be used to develop the system’s key computer-processing features. This will potentially enable real-time (live) information to be given to surgeons in the future whilst they are performing the procedure; and has the potential to make neurosurgery safer and more precise.
We are currently running a public engagement group that had its first meeting on 9th February 2022, providing valuable to our research team on this study.
We plan to meet 6 monthly to annually. If this is something you would like to be involved in please use the contact form. Similarly, if you would like more information about what the study entails from a patient perspective, please take a look at our study patient group pamphlet and our Patient Information Sheet (PIS).
Alternatively, feel free to contact us and one of the research team will get back to you.
News
Post overview:
- Focus: Support surgical imaging data analysis tasks and work on computational algorithms to help streamline data annotation
- Line manager: Tom Vercauteren
- Salary: Grade 5, £38,482 - £43,249 or Grade 6, £44,355 – £47,882 (max SP34) per annum including LWA depending on experience
- Duration: Fixed term contract until 30 Sep 2025

Two neurosurgical studies led by CAI4CAI researching novel, light based, imaging systems that can be used to greatly improve the efficacy of future neurosurgical procedures ran a successful day of patient involvement.

The 25th of September saw the latest meeting of “Science for Tomorrow’s Neurosurgery,” our now well established PPI group. As always, lots of exciting and valuable discussion with updates from Oscar on the (nearly complete!) NeuroHSI recruitment as well as Matt announcing the official opening of NeuroPPEYE phase 2!
Plain English Summary of the NeuroPPEye research project
Brain tumour surgery for low-grade glioma (LGG) improves patient outcome and survival but is very challenging. Even with the most advanced surgical techniques it remains difficult to determine tumour from healthy brain tissue. Advanced surgical methods can use light (fluorescence) created by the tumour to guide surgery. However, in the case of LGG, hardly any fluorescence light is visible, and each surgeon may judge the location and amount of light differently. Recent smart camera systems have the potential to enhance the surgeon’s vision to more reliably identify tumour and healthy brain structures. Hyperspectral imaging (HSI) is one of the most promising of such technologies providing rich information that is invisible to the human eye and could provide accurate and detailed measurements of tumour fluorescence. Using HSI, this study aims to develop objective surgical guidance to provide detailed real-time tissue margin information during surgery providing a step- change for treating LGG patients.
Scientific Abstract
Primary brain and central nervous system (CNS) cancers affect children and adults with 308,102 cases and 251,329 deaths registered globally in 2020 (Sung 2021). Global incidence of CNS cancer increased 17.3% between 1990-2016, causing 7.7 million disability-adjusted life years globally (Patel 2019).
HGGs and LGGs are the commonest CNS cancers, with LGGs accounting for 6.4% of all adult cases (Ostrom 2019). Despite LGGs typically being slow-growing, over 70% of them transform into higher- grade tumours or become aggressive within a decade (Jooma 2019). Median survival for LGG patients spans 5.6 to 13.3 years (Brown 2019).
Gross total resection (GTR) improves 5-year LGG survival rates from 60% to 90% when compared to subtotal resection. However, GTR (>96% tumour removal) is frequently not achieved because despite advanced techniques being available, surgeons are unable to clearly visualise the tumour and its boundaries in real-time during surgery
This project aims to improve patient survival by delivering a precise assistive tool for neurosurgeons performing LGG surgery. For HGGs, Protoporphyrin IX (PpIX) fluorescence-based imaging induced by 5-aminolevulinic acid (5-ALA) is the clinical standard for intraoperative surgical guidance. Yet this technique is non-quantitative and relies upon the surgeon’s subjective judgement to determine tumour margins. Furthermore, 5-ALA-PpIX fluorescence is of limited use in LGG resection because only 10-20% of LGGs show PpIX accumulation visible to the human eye.
Our innovation integrates advanced computational biophotonics with HSI integrated in a standard surgical microscope, enabling surgeons to quantitively assess LGG margins in real-time. This project builds on our leading expertise and previous development including a preliminary quantitative 5-ALA- PpIX benchtop imaging system (Xie 2017) and a contrast-agent-free intraoperative HSI with first-in- patient demonstration (Ebner 2021). On the software and algorithmic side, we build on the leading expertise in the team on the implementation of real-time computational biophotonics (Ravì 2019; Savoire 2012).
Contact
NeuroPPEye team
- 1 Lambeth Palace Road, London, SE1 7EU, UK
- Becket House 9th floor